
Among the specialty elastomers, polychloroprene [poly(2-chloro-1,3-butadiene)] is one of the most important with an annual consumption of nearly 300 000 tons worldwide. The first production was in 1932 by DuPont (“Dupree”, later “Neoprene”) and since then CR has an outstanding position due to its favorable combination of technical properties.
CR is used in different technical areas, mainly in the rubber industry (ca.61%), but is also important as a raw material for adhesives (solvent and water based, ca.33%) and has different latex applications (ca.6%) such as dipped articles (e.g. gloves), molded foam and improvement of bitumen.
The typical delivery form of CR is “chips”, a consequence of the special production process (see below). Application areas in the elastomer field are widely spread, such as molded goods, cables, transmission belts, conveyor belts, profiles etc.


CR is not characterized by one outstanding property, but its balance of properties is unique among the synthetic elastomers. It has:
Polychloroprene can be vulcanized by using various accelerator systems over a wide temperature range.
From the beginning until the 1960s, chloroprene was produced by the older “acetylene process”. This process has the disadvantages of being very energy-intensive and having high investment costs. The modern chloroprene process, which is now used by nearly all producers, is based on butadiene, which is readily available. Butadiene is converted into the monomer 2-chloro-butadiene-1,3 (chloroprene) via 3,4-di-chloro-butene-1.
In principle, it is possible to polymerize chloroprene by anionic, cationic and Ziegler-Natta catalysis techniques. However, because of product properties and economic considerations, free radical emulsion polymerization is used exclusively today. It is carried out on a commercial scale using both batch and continuous processes.
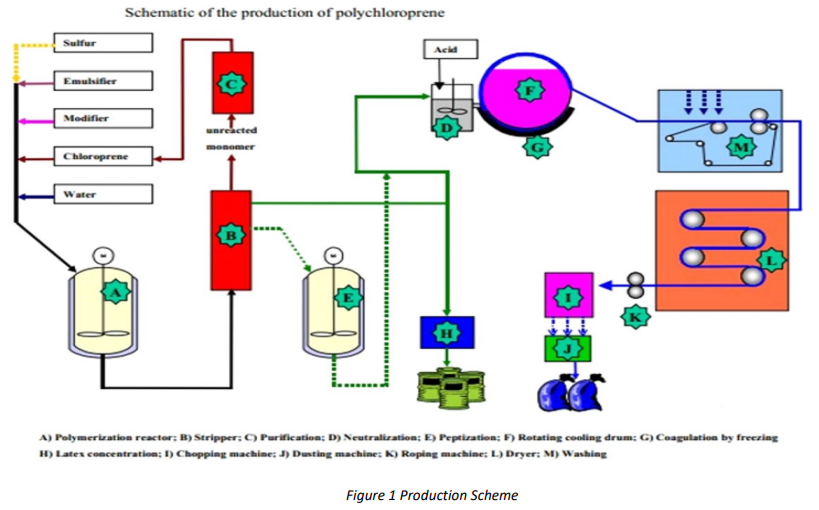
With the aid of radical initiators, chloroprene in the form of an aqueous emulsion is converted into homopolymers or, in the presence of comonomers into copolymers. The polymerization is stopped at the desired conversion by the addition of a short-stopping agent. The latex is freeze-coagulated on large, refrigerated revolving drums, from which it is drawn as a thin sheet. After washing and drying, the sheet is formed into a rope and then chopped to form the familiar chips or granules.
The basic polymerization scheme leads to incorporation of the monomer into a polymer consisting of different structural units (Figure 2):
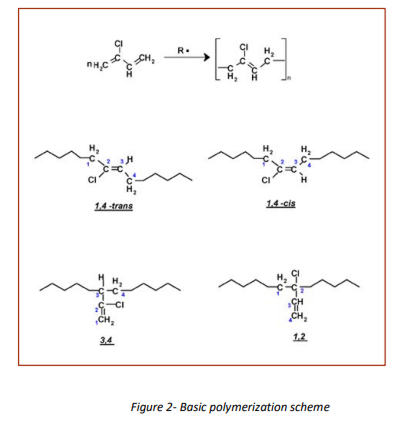
The physical, chemical and rheological properties of the different grades of commercial polychloroprene are dependent on the ability to change the molecular structure by changing polymerization conditions, e.g. polymerization temperature or monomer conversion, polymerization aids (comonomers, type and amount of molecular weight modifier and emulsifier) and conditions during finishing.
The high amount of trans-1, 4-units in the polymer (about 90 % at standard polymerization conditions) leads to synthetic rubber, which has crystallization as an inherent property.
Building a Sutainable Future for Your Organization and the Synthetic Rubber Industry
Download here

Address:
16360 Park Ten Place Suite 110
Houston, TX 77084
Contact Us:
+1 713 783 1703
info@iisrp.com